On February 11th, NBC News published a story by writers Kenzi Abou-Sabe, Yasmine Salam, and Cynthia McFadden titled “Can giving the ocean an antacid help curb climate change?,” describing ways to artificially induce the world’s oceans to capture and store more carbon dioxide.
Whether artificially altering the oceans chemistry is a worthwhile or valuable goal to fight climate change is unclear, what is fairly clear, it is impractical due to scale, and could possibly produce unintended negative consequences.
The NBC News story is perhaps best summed up in the first few paragraphs of the story:
Just over a year ago, Canadian oceanographer Will Burt was in Fairbanks, Alaska, teaching college students about the effects of global warming on marine life when a former colleague approached him about a startup seeking to use the ocean to remove carbon from the atmosphere.
“I didn’t have to think about it,” Burt said.
…
Burt works for Planetary Technologies, a Canadian startup that’s attempting to harness and accelerate that potential by adding antacid powder to the ocean.
The theory goes that by altering seawater chemistry, the ocean’s surface could absorb far more atmospheric carbon than it does naturally.
The premise is that with human intervention the world’s oceans could absorb more carbon dioxide (CO2), to be dissolved in them, in order to slow or reverse the growth of CO2 in the atmosphere. However, Burt and other researchers seem to misunderstand basic chemistry, which tell us that as the temperature of the oceans increase, CO2 will outgas from it. This has been known for decades, as seen in the figure below.
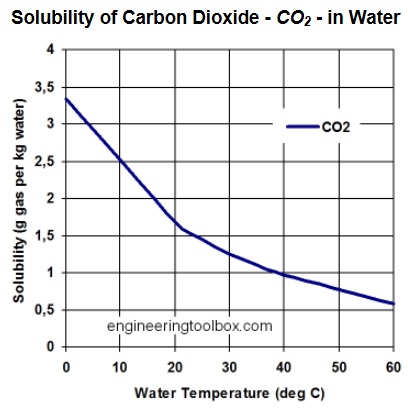
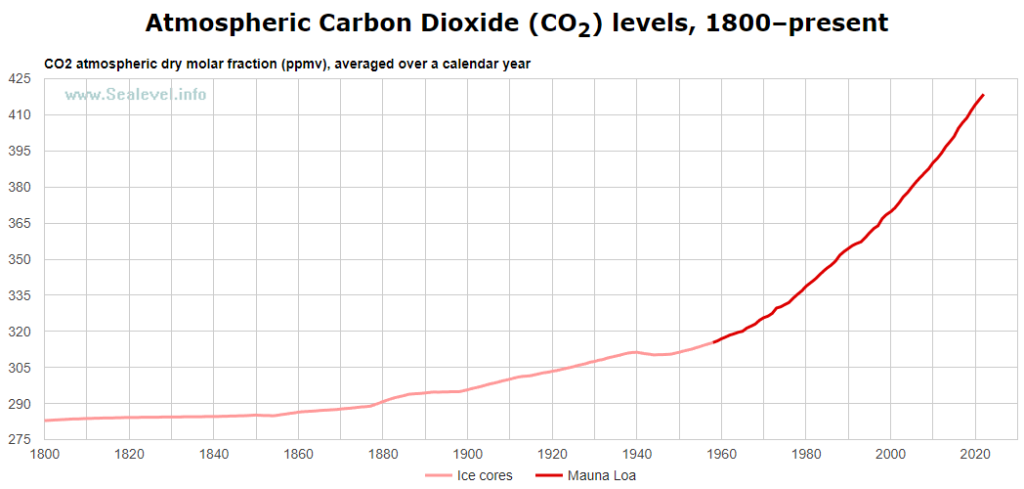
So, just like soda pop fizzes out more carbonation when it is warm vs. cold, the oceans outgas more CO2 as they warm. The Earth is modestly warming, and has been since about 1850. As water temperature increases with a warmer planet, CO2 solubility becomes less, and the oceans outgas more CO2. As seen in Figure 2, CO2 has been increasing in the atmosphere, partly due to human contributions, but also due to outgassing of CO2 from the oceans as the Earth warmed since the end of the Little Ice Age, which ended around1850.
Adding antacids won’t have any effect on the temperature dependent chemical process in the oceans, and these researchers will soon discover that their efforts would be miniscule in the big picture.
Of course, when Burt said “I didn’t have to think about it.” he was correct, and any logical person can see the folly of his quest just on the impossibly large scale alone, never mind the uphill battle against decreasing CO2 solubility and ocean outgassing as temperature increases.
And, have oceanographers like Burt given any thought whatsoever to dumping huge amounts of an antacid compound into the ocean? On one hand, we are told the oceans are fragile, on the other hand we are told by Burt and others they should be used for an experiment in industrial-scale antacid tests. The article says, “Planetary intends to recycle mine waste from a defunct asbestos mine in Quebec to produce pure magnesium hydroxide, which the company believes would help accelerate the ocean’s carbon uptake ability in the areas where it’s used.”
Dump asbestos mine waste in the ocean? Surely there must already be an environmental law against such a thing.
The idea that we ought to attempt to intentionally alter global ocean chemistry by dumping refined asbestos mine waste into them is wrong-headed. Even if it made sense, with no unintended consequences, the scale needed to appreciably reduce atmospheric CO2 is likely impossible.
















